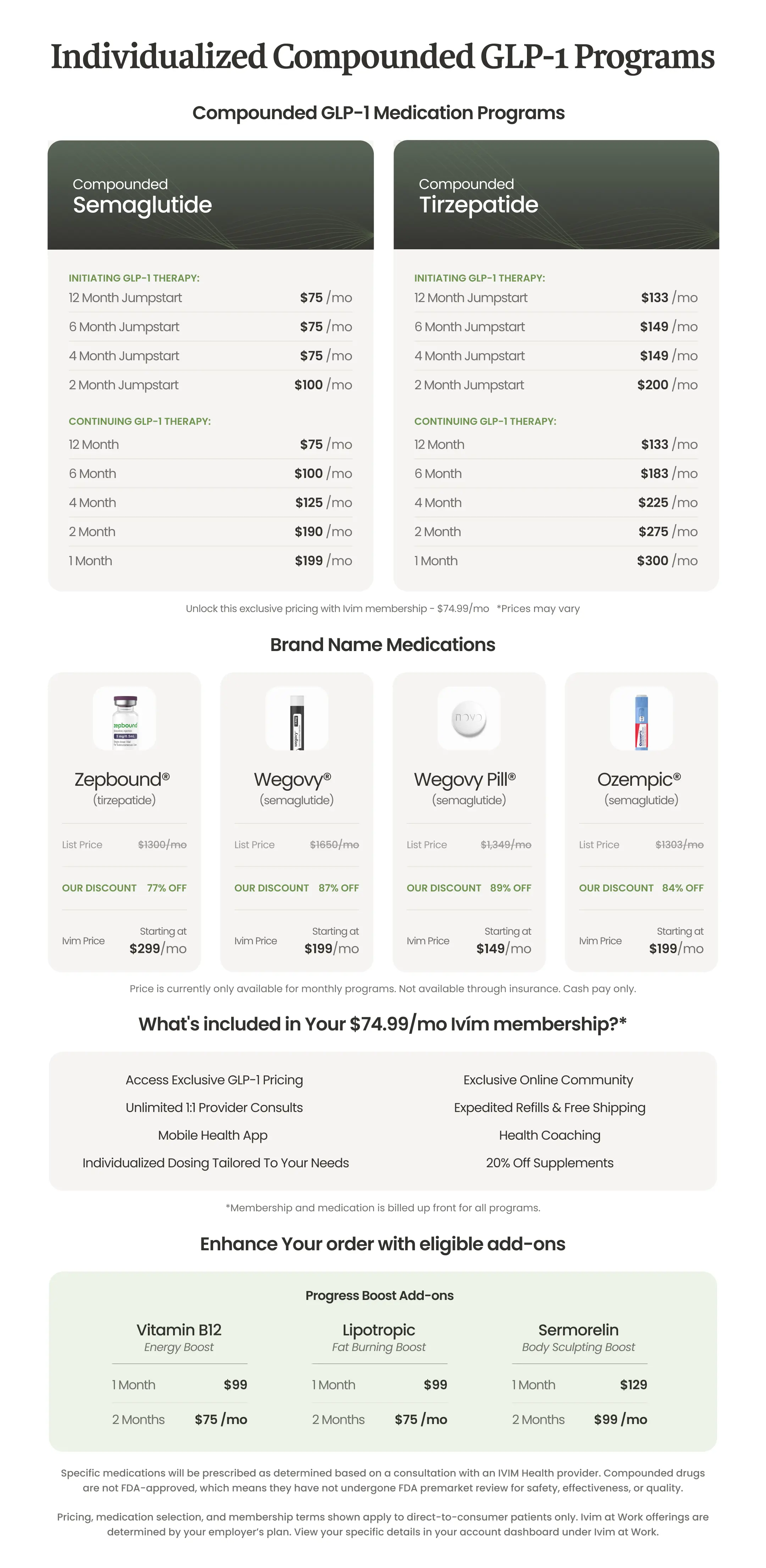
Individualized Weight Loss
*Based on Ivim’s published study, Ivim patients following the Ivim Protocol lost an average of 46% more weight over 68 weeks when compared to another published clinical GLP-1 trial that included a daily 500-calorie deficit, 150 minutes of weekly exercise, and in-person counseling every four weeks. Not a head-to-head study. RX required after clinical evaluation. This is a compounded medication and is not FDA-approved. Compounded drugs have not undergone FDA review for safety, efficacy, or quality. See Ivím website for full safety information. Results may vary.
Your Ivím GLP-1 ID Patient Journey
Here’s how it works:
What your individual program includes
Competitors
Medicine
ONLY
$850 value
coaching & community
$500 value
formulated supplements
Ivím Health App
What to Expect
Your Ivím GLP-1 ID Patient Journey
Understanding individualized compounded GLP-1 medications
Learn more about GLP-1s
-
Are the New GLP-1 Pills as Effective as Ozempic? An Obesity Doctor Explains
Most people I know have an opinion about GLP-1 medications. Fewer have accurate information. I sat down with Dr. Jessica Duncan, Chief Medical Officer at Ivim Health and a board-certified obesity medicine physician, to ask the questions worth actually asking. Start from […]
-
Ivim Health Welcomes FDA’s Long-Overdue Step Toward Regulated Peptide Access
Anthony Kantor, Dr. Taylor Kantor, and Dr. Jessica Duncan — We applaud today’s announcement from Secretary Kennedy on peptide reclassification. This is meaningful news for the patients we serve every day. For too long, Americans interested in peptide therapy have had limited options […]
-
Ivim Live Recap: Why Hydration Is the Foundation of Your GLP-1 Journey
At Ivim Health, we believe that a GLP-1 medication is only as powerful as the lifestyle foundation built around it. That’s the driving philosophy behind Ivim Live, our webinars where our functional health coaches go deep on […]
*Based on Ivim’s published study, Ivim patients following the Ivim Protocol lost an average of 46% more weight over 68 weeks when compared to another published clinical GLP-1 trial that included a daily 500-calorie deficit, 150 minutes of weekly exercise, and in-person counseling every four weeks. Not a head-to-head study. RX required after clinical evaluation. This is a compounded medication and is not FDA-approved. Compounded drugs have not undergone FDA review for safety, efficacy, or quality. See Ivím website for full safety information. Results may vary.
*Based on our internal data and individual patient outcomes may vary. This does not constitute a guarantee of specific results.
✝ These results include data from patients using both FDA-approved and compounded formulations.
Individualized virtual integrative medicine (IVIM): A clinical model for enhanced GLP-1 therapeutic outcomes